Midwest Pediatric Device Consortium
Who We Are
The Midwest Pediatric Device Consortium (MPDC) is an FDA funded hub of Ohio Children’s Hospitals including:
What We Do
DIRECT DEVICE FUNDING
Semiannual grant funding to aid in development and commercialization, ranging from $10,000 to $250,000.
CLINICAL TRIALS NETWORK
Consulting, advising and help implementing trials and studies needed for commercialization.
REAL WORLD EVIDENCE AND DATA
Accessing hospital data to inform market validation, expanded labeling and post-market surveillance.
PEDIATRIC LANDSCAPE
Oversight on pediatric projects being developed and cleared by the FDA for the use of children's hospitals, medical associations, developers, and investors.
We are committed to leveraging the expertise and resources of the consortium to expedite the development, production and distribution of pediatric medical devices. We do this through:
Grant funding ($10,000 to $250,000)
Coaching and education around pediatric medical device development
Access to industry leaders and partners
Access to clinicians and physicians
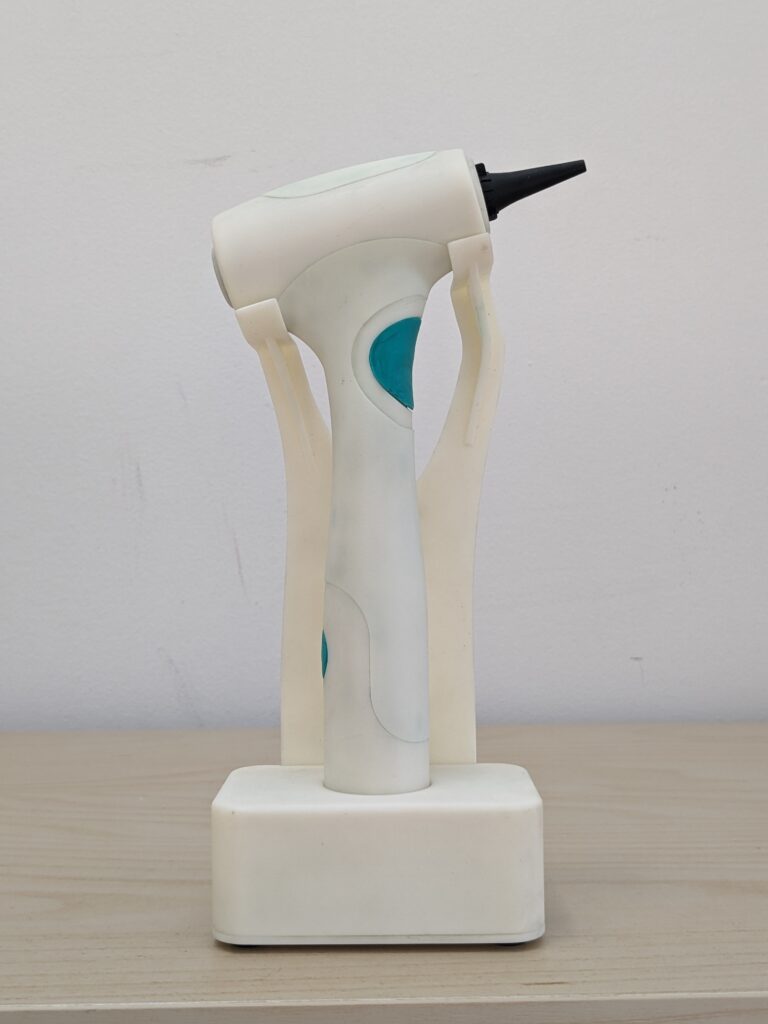
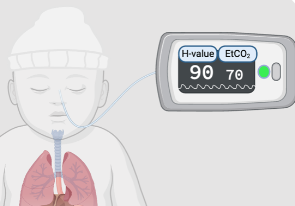
See Products We’ve Championed
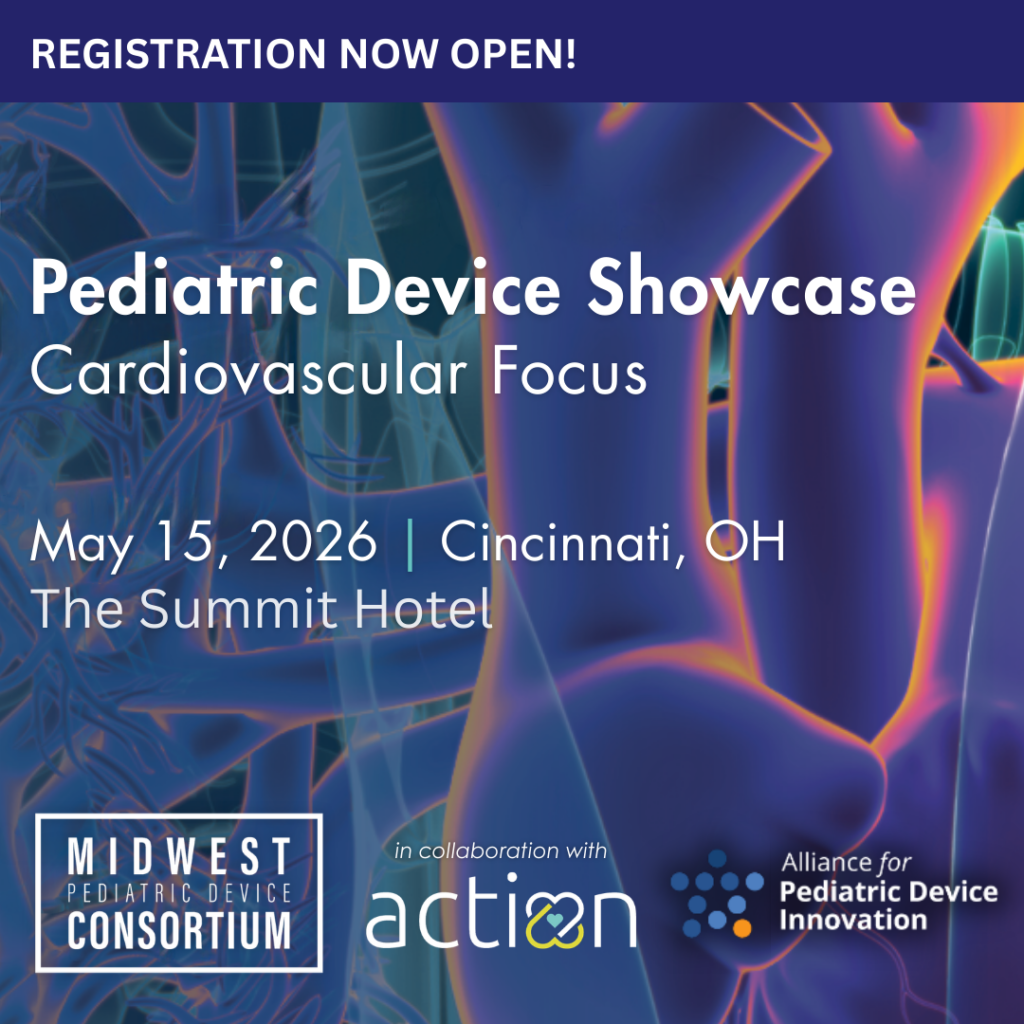
News & Events
Keep up to date on the latest grant updates, announcements, showcases, webinars and more.
Looking to get involved and support pediatrics?
Whether you’re an inventor, clinician, business, investor or pediatric advocate, we have a place here for you to get involved.
I AM: